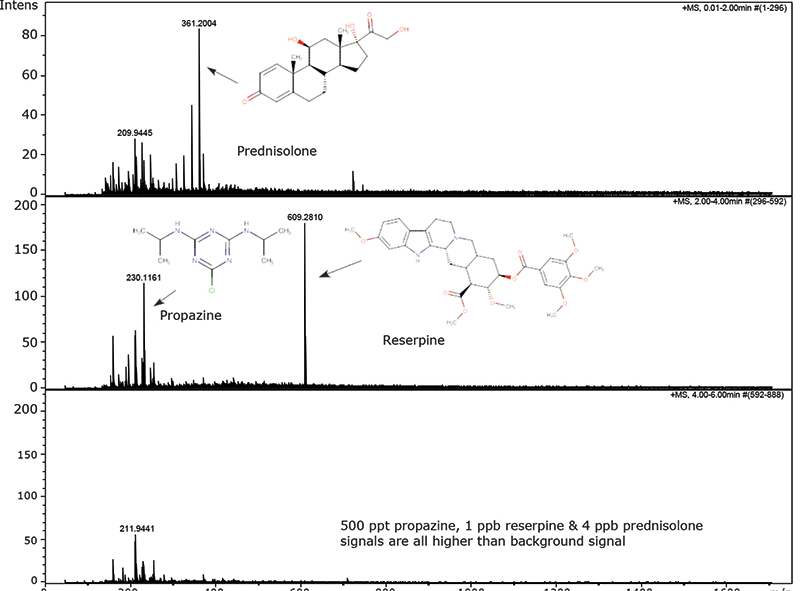
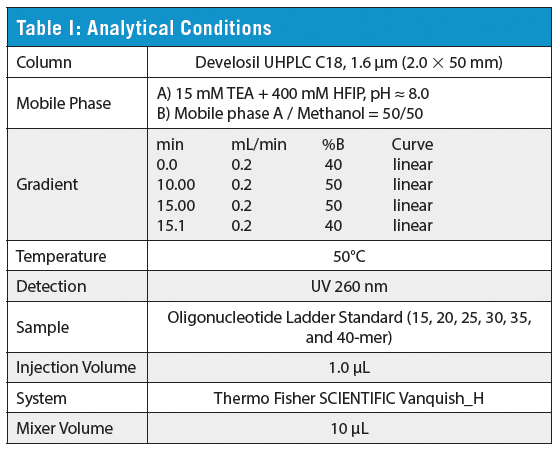
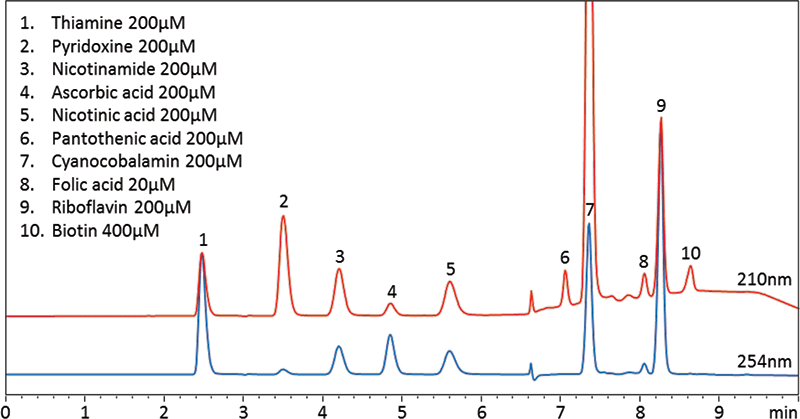
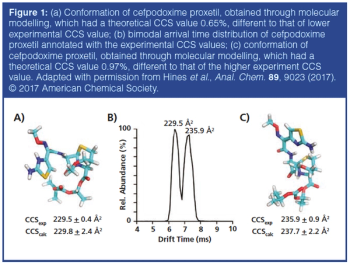
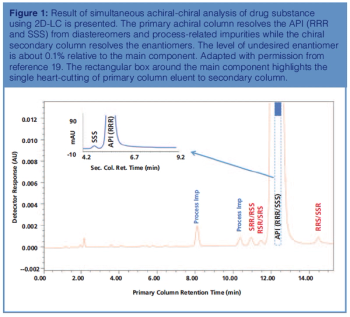
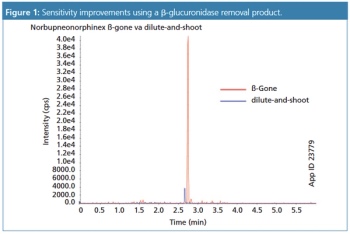
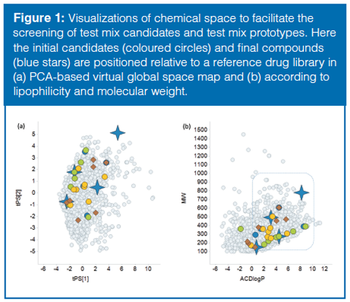
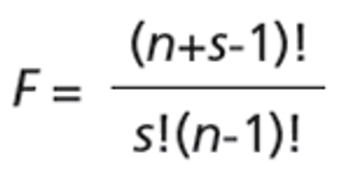This review article will give a general overview of the liquid chromatographic (LC) and gas chromatographic (GC) methods used by analytical laboratories for the detection and characterization of suspected illegal medicines and health products, including lifesaving drugs (antimicrobials and antimalarials), lifestyle drugs (erectile dysfunction drugs), and biotechnology drugs (doping peptides and skin-tanning peptides). Literature published from 2015 until early 2019 will be surveyed.