
Most analytical approaches used for the determination of low-molecular-weight noncovalent aggregates in protein pharmaceuticals suffer from important limitations. This first part of this two-part column series discusses those limitations.
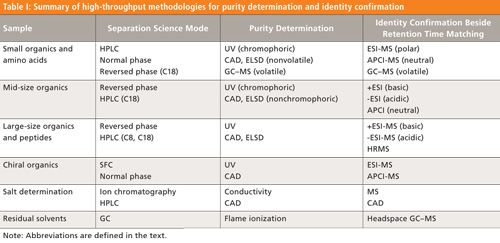

Most analytical approaches used for the determination of low-molecular-weight noncovalent aggregates in protein pharmaceuticals suffer from important limitations. This first part of this two-part column series discusses those limitations.

In this installment, we discuss fluorescence-detection size exclusion chromatography (FSEC) technology and its implications in protein characterization, specifically the development and characterization of biologics.

A discussion of the role of the gradient in separation by reversed-phase and ion-exchange HPLC, with data from two products to illustrate the key points

During the past year, LCGC examined current trends in the application of liquid chromatography (LC), and gas chromatography (GC), and related techniques in environmental, food, forensics, and pharmaceutical analysis. This article presents some developments made by separation scientists working in these application areas and offers insights into the current trends in each field.

The modern usage and specific applications of HILIC for two other major classes of analytes - glycopeptides and glycoproteins - are discussed.


Answers to common questions about HILIC, and an overview of its application to biopharmaceutical analysis.

LCGC recently spoke with Pauline Rudd of University College, Dublin, and The National Institute for Bioprocessing Research and Training (NIBRT) in Dublin, Ireland, about her work using ultrahigh-pressure liquid chromatography (UHPLC) for the characterization of protein glycosylation.

The use of a monolithic column reduces analysis time in an on-line biopharmaceutical HPLC analysis of granulocyte colony stimulating factor.

Recent advances in sampling techniques in the pharmaceutical industry sparked significant interest in applying improvements to extraction methods for greater analyte detection and quantitation.

A method using HR-MS systems to characterize the structures of metabolites is presented. The result is a general workflow for metabolism studies in drug discovery and development.

Click here to view the complete E-Separation Solutions newsletter from August 22, 2013.
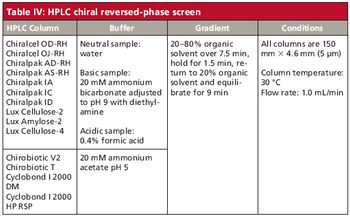
A set of extended chiral separation screens using HPLC and SFC has been tailored to meet the specific and changing needs of customers in a pharmaceutical development setting.

HPLC method development can be accelerated by using three distinct method templates of increasing complexity.

Pharmaceutical Analysis Technology Forum

Click here to view the complete E-Separation Solutions newsletter from May 23, 2013.

This article highlights some selected examples of the power of liquid chromatography combined with mass spectrometry (LC–MS) in the development of protein biopharmaceuticals.

The determination of genotoxic impurities (GIs) in drug substances and pharmaceutical products is an emerging topic in pharmaceutical quality control. GIs are intermediates or reactants in the synthetic pathway of a drug substance and should be monitored at ppm (?g/g drug substance) or even ppb (ng/g) levels. This is several orders of magnitude lower than in classical impurity analysis (0.05% or 500 ppm level) or in residual solvent analysis. Analytical methods for the determination of GIs include gas chromatography (GC) and liquid chromatography (LC), both often combined with mass spectrometry (MS) detection. Some typical examples of GIs trace analysis using GC and LC are presented. The potential of on-line reaction monitoring is also discussed.

This article provides an overview of the most recent advances in the field of chiral and achiral separations in SFC. This involves research focused on the most critical parameters in SFC separations, but also on practical issues such as the serial coupling of columns.

Hydrophilic interaction liquid chromatography (HILIC) has recently become more important, particularly for the analysis of polar drugs, metabolites and biologically relevant compounds in glycomics, proteomics, metabolomics and clinical analysis. HILIC makes it possible to increase the retention of polar compounds, achieve orthogonal selectivity and increase mass spectrometry (MS) sensitivity, compared with reversed-phase liquid chromatography. This article discusses the advantages and limitations of HILIC in a variety of practical applications in the pharmaceutical industry.

A new type of method development that uses modeling to find the "best" separation for high performance liquid chromatography (HPLC) was investigated and principles of Quality by Design (QbD) were followed when planning the investigation. The process delivered precise results and the method was able be transferred to a routine quality control (QC) laboratory.

In biopharmaceutical development the stability of biological molecules in drug formulations is important. This article describes three different approaches to the measurement and quantification of aggregates in protein solutions, and highlights the range of information that each technique can provide about the sample.

How to use these tools to develop an efficient and robust process

By achieving selectivity in other portions of the analytical cycle, we can lower the burden on sample preparation.

Detailed examples of light-scattering techniques are presented as well as how their performance compares to MS and other methods.