
LCGC North America
Click the title above to open the LCGC North America May 2018 regular issue, Vol 36 No 5, in an interactive PDF format.

LCGC North America
Click the title above to open the LCGC North America May 2018 regular issue, Vol 36 No 5, in an interactive PDF format.

Special Issues
Click the title above to open the May 2018 issue of Current Trends in Mass Spectrometry, Volume 16, Number 2, in an interactive PDF format.

The Column
Agilent Technologies Inc. and Imperial College London have signed a strategic scientific collaboration agreement that will see a broad range of Agilent instruments installed in a sponsored measurement suite to be housed within Imperial’s Molecular Sciences Research Hub.

The Column
Researchers from the University of Minnesota have investigated the salivary proteomics of healthy dogs using offline high pH reversed-phase LC fractionation and nano LC–MS/MS.

The Column
Incognito focuses on a big challenge in regulated analytical chemistry laboratories.
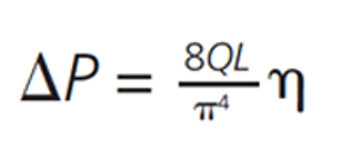
The Column
This article introduces the development of an automated and versatile technique for solution viscosity determination of a wide range of polymeric materials in different solvents. Sample preparation is a tedious and error-prone process in viscosity determinations of polymeric materials, especially when elevated temperature or when hazardous solvents are required. This new approach automates sample preparation and injection to the viscometer detector, with benefits in efficiency and safety as well as in analysis precision. The dissolution conditions were studied and optimized to reduce thermal and oxidative degradation, which are detrimental to the accuracy of the observed viscosity.

The Column
Click the title above to open The Column April 2018 Europe & Asia issue, Volume 14, Number 4, in an interactive PDF format.

The Column
Click the title above to open The Column April 2018 North American issue, Volume 14, Number 4, in an interactive PDF format.

What is the chemistry of this phase? What are the mechanisms of interaction with the analyte and hence how is retention and selectivity gained from this phase? How can we troubleshoot separation problems or develop suitable methods without a good knowledge of the bonded phase chemistry?


I do not remember the application, but I remember very clearly Professor McNair telling us that soil is one of the most challenging sample matrices, if not the toughest, from which to perform analytical determinations. Sources indicate the composition of soil ideal for growing plants to be 25% air, 25% water, 45% minerals, and 5% organic matter. That does not seem like a daunting makeup, but the reality is that the relative proportion of the constituents can vary dramatically.

LCGC North America
Our annual review of new liquid chromatography columns and accessories, including columns for reversed-phase, HILIC, chiral, SEC, GPC, ion-exchange, and SFC separations of small and large molecules.

LCGC North America
In part II of this series, we explore how curve resolution methods advance purity assessments and illustrate one of the most popular techniques, which adds a powerful tool to the chromatographer’s arsenal.

LCGC North America
Ligands capable of undergoing electrostatic interactions can add an extra dimension to separations of ionizable compounds.
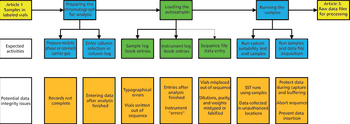
LCGC North America
Part II of this series on practical perspectives on data integrity focuses on instrument setup, system suitability test samples, and data acquisition.
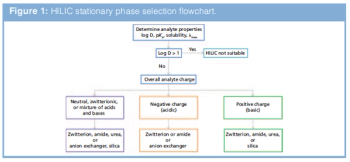
LCGC Europe
Tips on how to select a hydrophilic interaction chromatography (HILIC) stationary phase.

LCGC Europe
The Chromatographic Society and Joint Pharmaceutical Analysis Group have announced a one‑day meeting titled From Cradle to Grave: The Chromatographic (Analytical) Method Life Cycle. The meeting will be held at Burlington House, London, UK, on Thursday 10 May 2018.

LCGC Europe
Click the title above to open the LCGC Europe April 2018 regular issue, Vol 31, No 4, in an interactive PDF format.

LCGC North America
Click the title above to open the LCGC North America April 2018 regular issue, Vol 36 No 4, in an interactive PDF format.


Our new sister publication, Cannabis Science and Technology (CST), launched in March 2018.

The Column
This article introduces the development of an automated and versatile technique for solution viscosity determination of a wide range of polymeric materials in different solvents.


The Column
Click the title above to open The Column March 2018 Europe & Asia issue, Volume 14, Number 3, in an interactive PDF format.

The Column
Click the title above to open The Column March 2018 North American issue, Volume 14, Number 3, in an interactive PDF format.

The overriding majority of articles on problems with the technical transfer of HPLC methods ultimately focus on differences between HPLC dwell volumes. However, as the title suggests, there are many more issues which can cause problems in the transfer of HPLC methods, and I wanted to highlight some common issues that come across my desk, in the hope that it will help you avoid these problems in your own practice.

Agilent Technologies has signed a definitive agreement to acquire privately held Advanced Analytical Technologies, Inc.

Several years ago, I would have held the stance that environmental analysis was fairly boring. How complicated can water be? I am not ashamed to say that was a naïve view. It is clear from our research and related research by others on similar topics that much more work in these areas is needed. Standard methods cannot solely accommodate the growing list of targets and the multitude of unknowns associated with complex samples taken from the interface between the petroleum industry and the environment.

Special Issues
A novel “dilute-and-shoot” LC–MS/MS method is described for the analysis of “bath salts” sold as “legal” highs, including mitragynine and nine synthetic cathinones, in urine.

Special Issues
Novel ionization processes provide gas-phase ions of a wide variety of materials using MS. These simple and sensitive methods operate from solution or a solid matrix. Both manual and automated platforms are described that allow rapid switching between the ionization methods of MAI, SAI, vSAI, and conventional ESI.