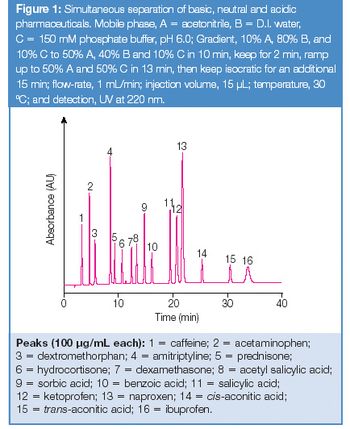
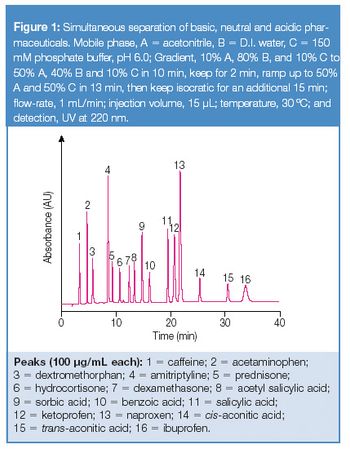
Paracetamol is a major ingredient in numerous medications due to its analgesic and antipyretic properties. During its synthesis (Figure 1), a total of ten process-related impurities are observed. Several HPLC applications have been developed for the monitoring of these impurities (1, 2), including the European Pharmacopoeia which has adopted an isocratic HPLC method using a silica-based C8 column with 5 μm particle size, requiring a run time of 45 min (3). By using a gradient method and standard HPLC instrumentation, the analysis can be reduced to 7 min (4).